Multiple Choice
Identify the
choice that best completes the statement or answers the question.
|
|
|
1.
|
In the reaction SO 2Cl 2 
SO 2+Cl 2 heat is evolved. What happens when chlorine (Cl 2) is added
to the equilibrium mixture at constant volume? a. | The temperature of the system increases. | b. | The temperature of
the system decreases. | c. | More chlorine is produced. | d. | The temperature
remains unaffected. |
|
|
|
2.
|
A(n) _____ solution contains the maximum amount of dissolved solute for a
given amount of solvent.
a. | saturated | c. | suspended | b. | supersaturated | d. | unsaturated |
|
|
|
3.
|
Unlike in an ideal gas, in a real gas
a. | all particles move in the same direction. | b. | all particles have
the same kinetic energy. | c. | the particles cannot
diffuse. | d. | the particles can exert attractive forces on each
other. |
|
|
|
4.
|
The equilibrium constant for this reaction at 700.0 K is 0.44. 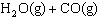 <-> <-> 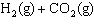 What is the
concentration of carbon dioxide gas, if [H2O] = 0.16 mol/L, [CO2] = 0.15 mol/L
and [H2] = 0.14 mol/L? a. | 0.0014 mol/L | c. | 0.16 mol/L | b. | 0.075 | d. | 0.75 mol/L |
|
|
|
5.
|
Water running over soil will cause:
a. | chemical weather | c. | Global warming | b. | Erosion | d. | Adhesion |
|
|
|
6.
|
A(n) _____ solution contains more dissolved solute than a saturated solution at
the same temperature.
a. | saturated | c. | suspended | b. | supersaturated | d. | unsaturated |
|
|
|
7.
|
Which of the following statements about a catalyst is true?
a. | A catalyst can initiate a reaction. | b. | A catalyst can accelerate a
reaction. | c. | A catalyst can be consumed during a reaction. | d. | A catalyst can be
changed during a reaction. |
|
|
|
8.
|
What volume of oxygen is needed to react with solid sulfur to form 6.20 L of
sulfur dioxide?
a. | 6.20 L | c. | 12.4 L | b. | 7.20 L | d. | 99.2 L |
|
|
|
9.
|
According to Le Chatelier’s principle, when the volume is increased, the
equilibrium shifts to the right for only one of the following. For which of the following reversible
reactions is this true?
|
|
|
10.
|
Which substance has the most space between its particles?
a. | H2O(g) | c. | H2O(s) | b. | H2O(l) | d. | all have the same space
|
|
|
|
11.
|
The __________ law of thermodynamics states that spontaneous processes always
proceed in such a way that the entropy of the universe increases.
a. | first | c. | third | b. | second | d. | fourth |
|
|
|
12.
|
If a collision between molecules is very gentle, the molecules are
a. | more likely to be oriented favorably. | b. | less likely to be oriented
favorably. | c. | likely to react. | d. | likely to rebound without
reacting. |
|
|
|
13.
|
A solution that contains equal concentrations of hydrogen and hydroxide ions is
_____.
a. | an acid | c. | neutral | b. | a base | d. | ionized |
|
|
|
14.
|
A substance that does not dissolve in a solvent is said to be:
a. | insoluble | c. | miscible | b. | immiscible | d. | soluble |
|
|
|
15.
|
More solute can be dissolved in a _____ solution:
a. | saturated | c. | suspended | b. | supersaturated | d. | unsaturated |
|
|
|
16.
|
Which system has the lowest entropy?
a. | Ice | b. | steam | c. | liquid water at room
temperature | d. | boiling water |
|
|
|
17.
|
What changes when a gas is compressed?
a. | mass | c. | density | b. | moles | d. | all of the
above |
|
|
|
18.
|
Which of the following factors will NOT change the concentration of ammonia
(NH 3) in the reaction? 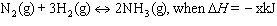 ? a. | Decrease in the volume of N2. | c. | Decrease in
pressure. | b. | Increase in the amount of catalyst. | d. | Decrease in
temperature. |
|
|
|
19.
|
In an exothermic reaction, equilibrium shifts _____ when temperature is
raised.
a. | to the left | c. | to the center | b. | to the right | d. | none |
|
|
|
20.
|
Which substance can dissolve only 40 g in 60 °C
water? 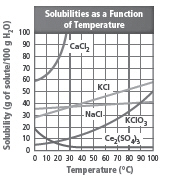 a. | NaCl | c. | KCl | b. | KClO3 | d. | Ce2(SO4)3 |
|
|
|
21.
|
A very high value of the
equilibrium constant for a reaction indicates that
a. | equilibrium is reached
slowly. | c. | reactants are
favored. | b. | equilibrium has been reached.
| d. | products are favored. |
|
|
|
22.
|
If the temperature of the
equilibrium system X + Y « XY + 25
kJ decreases,
a. | the concentrations of reactants and
products do not change. | b. | [X] decreases and [XY]
decreases. | c. | [X] decreases and [XY]
increases. | d. | [X] increases and [XY]
decreases.
|
|
|
|
23.
|
Two liquids that can be mixed together but separate shortly after are:
a. | immiscible | c. | miscible | b. | insoluble | d. | soluble |
|
|
|
24.
|
How much heat is evolved from 54.0 g glucose
(C 6H 12O 6), according to the equation for calculating
heat? 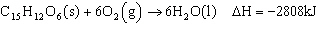 a. | 0.842 kJ | c. | 84.2 kJ | b. | 8.42 kJ | d. | 842 kJ |
|
|
|
25.
|
Calculate pH of an aqueous solution of hydrochloric acid. Given the hydrogen ion
concentration is 8.75 × 10–9 M.
|
|
|
26.
|
When ice melts to form liquid water it’s enthalpy _____ and its entropy
_____.
a. | increases, decreases | b. | decreases, decreases | c. | decreases,
increases | d. | increases, increases |
|
|
|
27.
|
When the volume of a gas decreases its pressure will ___ under constant
temperature.
a. | increases. | b. | decreases. | c. | remains
constant. | d. | It is impossible to tell because all gases are
different. |
|
|
|
28.
|
Which gas law has constant gas and volume? 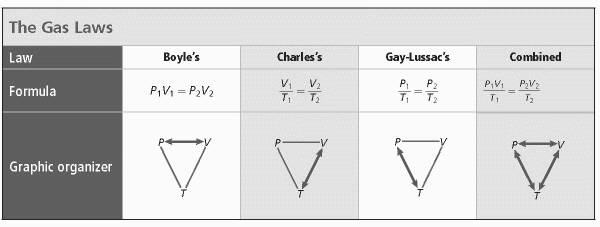 a. | Boyle’s | c. | Combined | b. | Charles’s | d. | Gay-Lussac’s |
|
|
|
29.
|
When temperature is lowered, pressure will _______ because _____.
a. | lower, particles slow down and collide less frequently with container
walls. | c. | lower, particles slow down and decrease the volume of the
container. | b. | lower, partilcles speed up and collide more frequently with container
walls. | d. | increase, particles
speed up and collide more frequently with container walls. |
|
|
|
30.
|
Which of the following is true of particles of a gas?
a. | attract each other but do not collide. | b. | repel each other and
collide. | c. | neither attract nor repel each other but collide. | d. | neither attract nor
repel each other and do not collide. |
|
|
|
31.
|
Based on the figure below, which of the following statements is true? 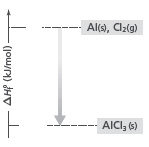 a. | The formation of AlCl3 began at 0.0oC. | c. | The final
temperature of the products was -704oC. | b. | The final temperature of the reactants was
-704oC. | d. | The
formation of AlCl3 releases energy. |
|
|
|
32.
|
What is happening to the energy of the universe in following
image?  a. | not enough information to tell | b. | decreasing | c. | staying the
same | d. | increasing |
|
|
|
33.
|
Keq ___ 1: Reactants are favored at equilibrium.
|
|
|
34.
|
What is the equation for calculating heat?
|
|
|
35.
|
The property of water that describes water sticking to a tree branch is:
a. | cohesion | c. | adhesion | b. | anomaly | d. | polarity |
|
|
|
36.
|
The _____ law of thermodynamics states that energy is neither created nor
destroyed.
a. | first | c. | third | b. | second | d. | fourth |
|
|
|
37.
|
A basic solution contains more _____ ions than hydrogen.
a. | oxygen | c. | hydroxide | b. | nitrogen | d. | sulfide |
|
|
|
38.
|
A _____ is produced when a base accepts a hydrogen ion from an acid.
a. | conjugate acid | c. | acid | b. | conjugate base | d. | base |
|
|
|
39.
|
In the Bronsted-Lowry model of acids and bases, a(n) _____ is a hydrogen donor
and a(n) _____ is a hydrogen acceptor.
a. | acid, base | c. | conjugate acid, conjugate base | b. | base,
acid | d. | conjugate base,
conjugate acid |
|
|
|
40.
|
What is the pH of blood, given the hydrogen ion concentration is 4.0 × 10-8 M?
|
|
|
41.
|
A collision requires _____ to be effective.
a. | only enough energy | b. | favorable orientation | c. | enough energy and
favorable orientation | d. | a reaction
mechanism |
|
|
|
42.
|
In an endothermic reaction, equilibrium shifts _____ when temperature is
lowered.
a. | to the left | c. | to the center | b. | to the right | d. | none |
|
|
|
43.
|
What is the conjugate acid in the forward reaction? 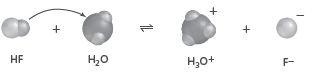 a. | HF | c. | H2O | b. | H3O+ | d. | 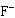 |
|
|
|
44.
|
Keq ___ 1: Products are favored at equilibrium.
|
|
|
45.
|
What unknown quantity is
calculated after performing a titration?
a. | concentration | c. | volume | b. | density | d. | mass |
|
|
|
46.
|
According to Gay-Lussac’s law:
a. | pressure is inversely proportional to volume at constant
temperature. | b. | pressure is directly proportional to temperature at constant
volume. | c. | volume is inversely proportional to temperature at constant
pressure. | d. | volume is directly proportional to temperature at constant
pressure. |
|
|
|
47.
|
Of all the water on earth the majority is:
a. | Salt water | c. | Liquid Water | b. | Water vapor | d. | Solid Ice |
|
|
|
48.
|
A _____ reaction is a chemical reaction that can occur in both the forward and
reverse directions.
a. | complete | c. | reversible | b. | forward | d. | incomplete |
|
|
|
49.
|
A decrease in the concentration of reactants causes the rate of the _____
reaction to slow.
a. | complete | c. | reverse | b. | forward | d. | incomplete |
|
|
|
50.
|
Which of the following is a measure of the disorder in a system?
a. | enthalpy | b. | entropy | c. | temperature | d. | free energy |
|